STUDY ON MICROBIOME and METABOLOME of ASIANS

Forty trillions of bacterial cells consisting of more than a hundred species of microbes inhabit digestive tract of human being. This microbial community called “gut microbiota” utilizes unabsorbed nutrients from consumed diets in the colon and affects host health through fermentative production of a variety of metabolites. Therefore, gut microbiota can be considered hidden organ and attracts much interest in medical and food sciences. Notably, we have attracted interest in gut microbiota of Asian people with plenty of dietary cultures and established international consortium called "Asian Microbiome Project(AMP)” to collect and share the information about the link among diets, gut microbiome, and health in each population. Thus far, we have conducted four phases study. In Phase I, we investigated gut microbiome of school-age children of five countries as a pilot study. In Phase II, we built up basal database of gut microbiota of Asian people covering from newborns to elderlies. In Phase III, we looked into the interaction of foods and gut microbiota in more depth in certain areas, namely Leyte Island in Philippines and Bangkok and Buriram in Thailand. In Phase IV, we focused on the association of gut microbiome with lifestyle diseases, particularly obesity and type 2 diabetes. As a result, we have gained insights in the following six points. (1) There are two types of gut microbiome in Asians. (2) The gut microbiome mirrors dietary habits in each country. (3) Among Asians, Japanese have unique gut microbiome structure such as prominently high abundance of Bifidobacterium. (4) In developing area, structure and function of gut microbiome are being changed under the modernization of diets. Notably, loss of classical type of dietary habit mainly consisting of crops and vegetables decreases fermentative production of short chain fatty acids which have indispensable role to maintain host metabolic and energy homeostasis. (5) In Japanese study, we found that high consumption of fat and alcohol induces dysbiosis. (6) Asian traditional foods contain ingredients having capabilities of conditioning of gut microbiome. Taken together, although Asian microbiome reflects goodness of Asian traditional foods, the fact that the introduction of modern diets have started remodeling Asian gut microbiome warrants retrospective studies to close up benefits of Asian traditional foods.
Phase-I study: Analysis of 303 school-age children from 10 cities of 5 countries in Asia[Paper published in Scientific Report, 2015, 5, 8397(ref.1)
We analyzed fecal microbiome of school children aged from 7 y to 11 y, who were expected to have dietary habit representing food culture of each country. We chose five countries, namely Japan, China, Taiwan, Indonesia, and Thailand, and investigate gut microbial composition of children living in urban and rural cites (n = 25 to 45 in each site). We performed random forest analysis that is a machine learning program by using the obtained microbial composition data. The result interestingly showed that the children in Japan, China, and Indonesia have a distinct microbiota whose origin of country can be identify at high accuracy by the computer program. On the other hand, children in Taiwan and Thailand showed mixed types of China and Japan and China and Indonesia, respectively.(Fig.1)。
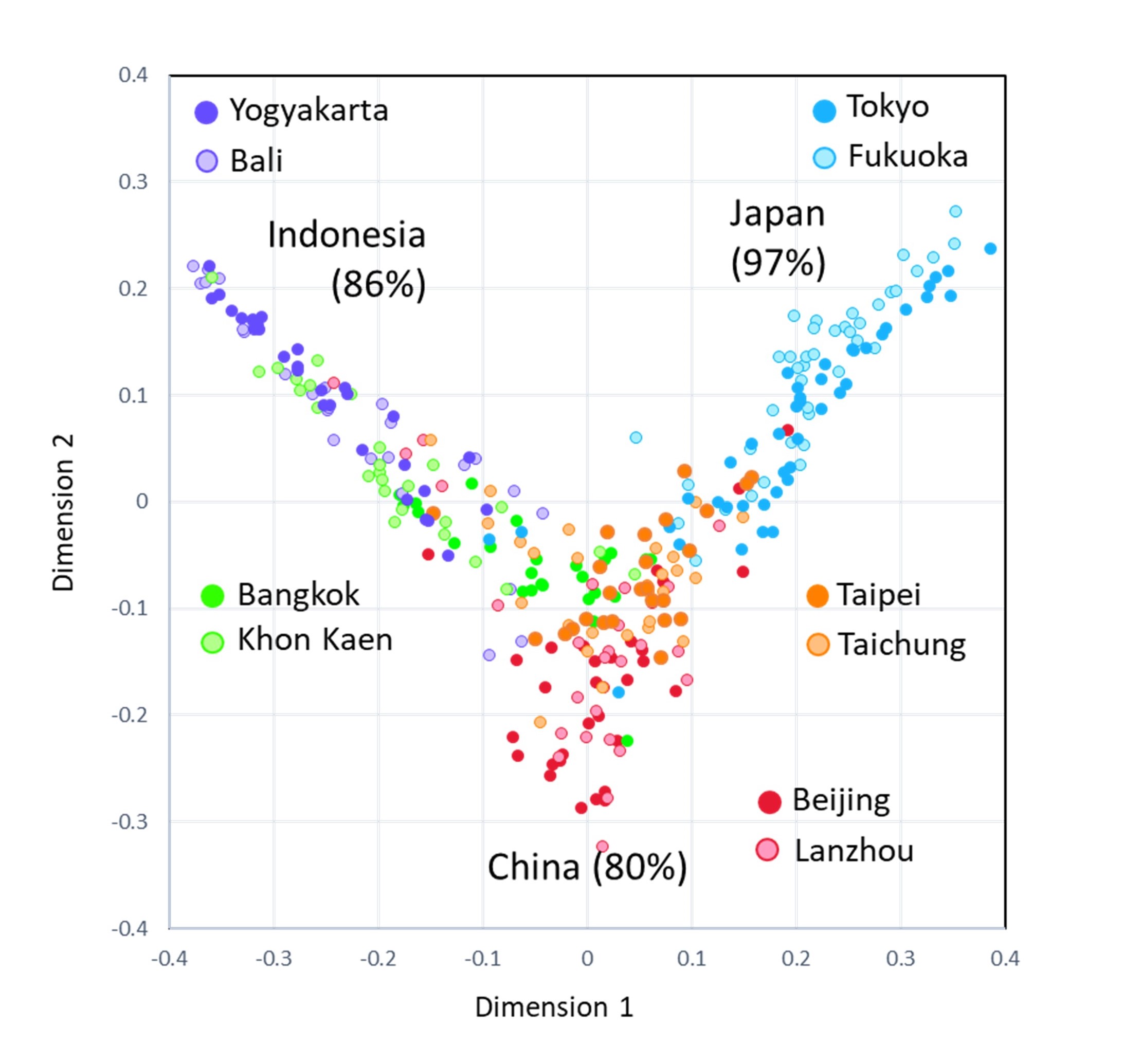
Fig.1.Random forest analysis of gut microbiota of 303 school-age children from 10 cities from 5 countries in Asia.
The gut bacterial composition data of 303 children was applied to a machine learning program (random forest analysis) and an algorithm to identify the country from which the sample was originated was established. It was able to identify the origin of country at 97% accuracy for Japanese and 86% for Indonesian, and 80% for Chinese, indicating most of children in these countries have microbiota unique to each country.
Principal component analysis of these 303 children's gut microbiota at bacteria family level segregated two different characteristic groups. One characterized by high abundance of Bacteroidaceae and Bifidobacteriaceae (BB-type) mainly consisted of children in Japan, Taiwan, China, and Bangkok in Thailand and the other one characterized by high abundance of Prevotellaceae (P-type) mainly consisted of children in Indonesia and Khon Kaen in Thailand (Fig.2). Particularly, 83 out of 84 Japanese children carried BB-type microbiota. Japanese children interestingly showed characteristic properties in terms of low alpha diversity (diversity within a sample)(Fig.3) and low beta diversity (diversity across samples)(Fig.4), suggesting the presence of factors strongly shaping monotonous BB-type microbiota in Japanese children. Maybe, highly hygienic controlled foods causes low diverse microbiota. The influence of this unique microbiota on the health and growth of Japanese children should be carefully monitored toward the future.
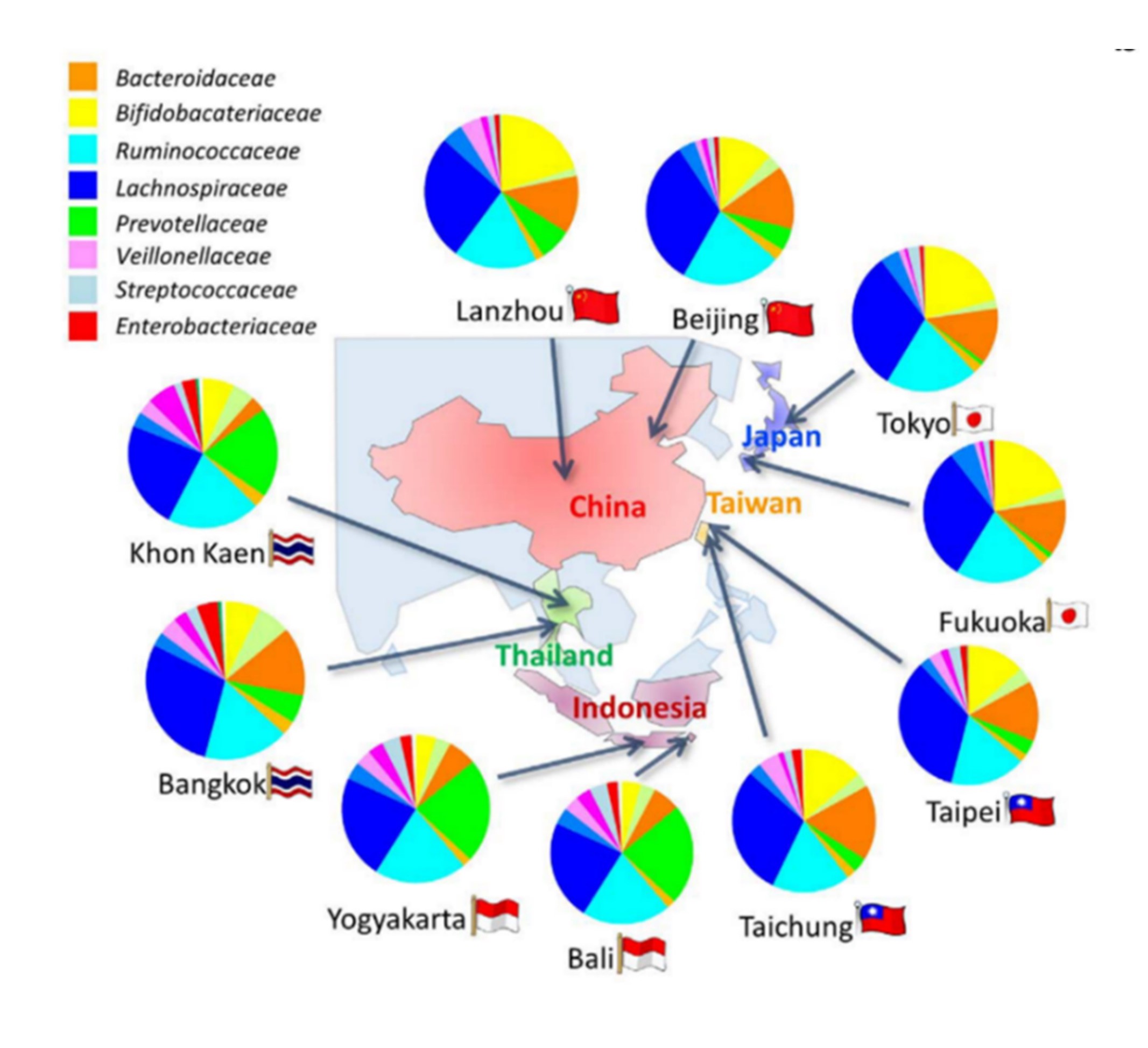
Fig.2. Bacteria composition at family level in feces of 303 children from 10 cities of 5 countries.
Children in south-east Asia were highly colonized by Prevotellaceae (Green), while children in east Asia were highly colonized by Bifidobacteriaceae (Yellow) and Bacteroidacea (Orange). Particularly, Japanese children from both Tokyo and Fukuoka are highly colonized by Bifidobacteriaceae.
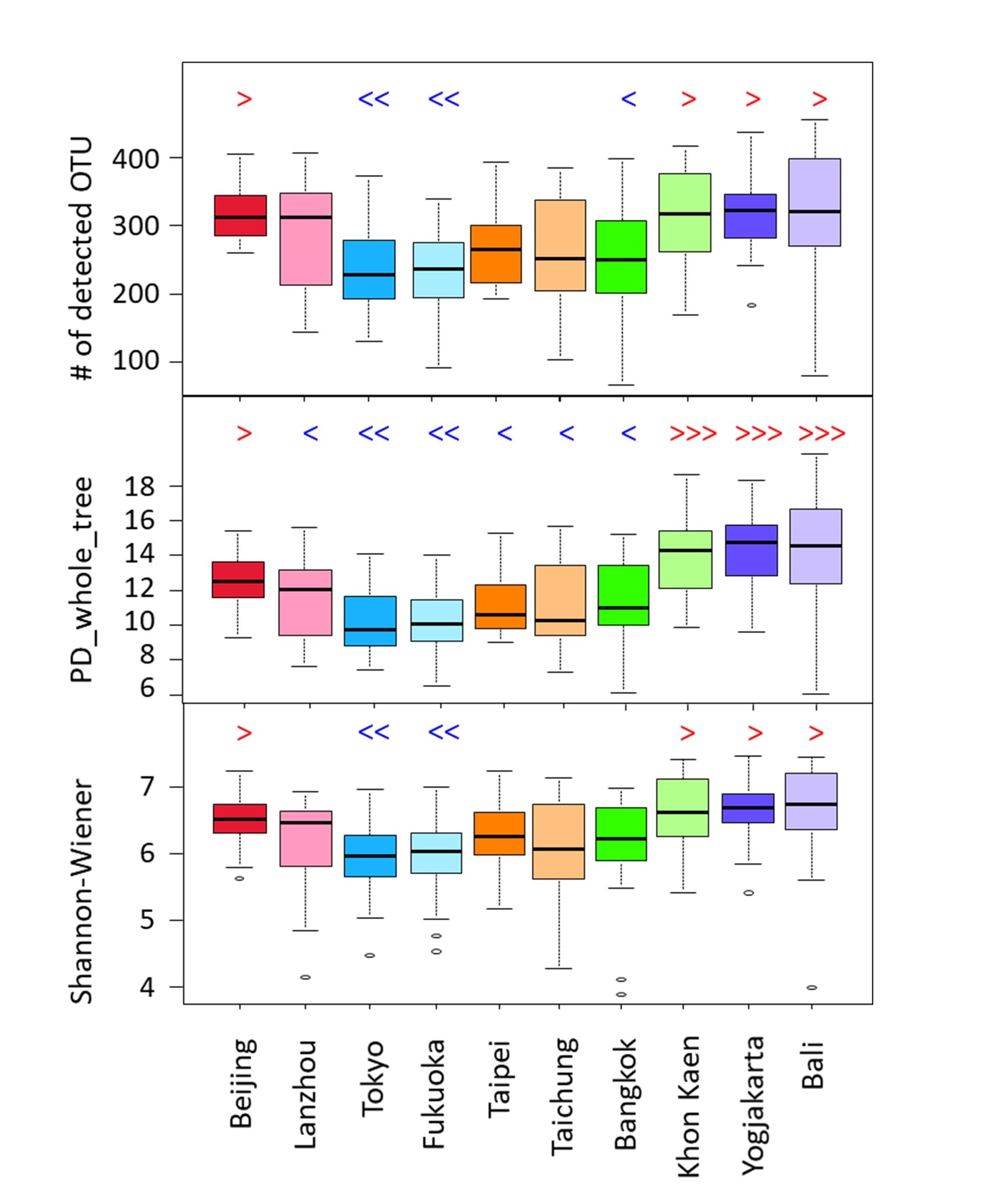
Fig.3. Alpha diversity of gut microbiota in children from 10 cities of 5 countries in Asia.
Japanese children from both Tokyo and Fukuoka showed significantly lower values in all three examined alpha diversity indices, indicating the presence of lower number of bacteria species in Japanese children compared to children in other countries.
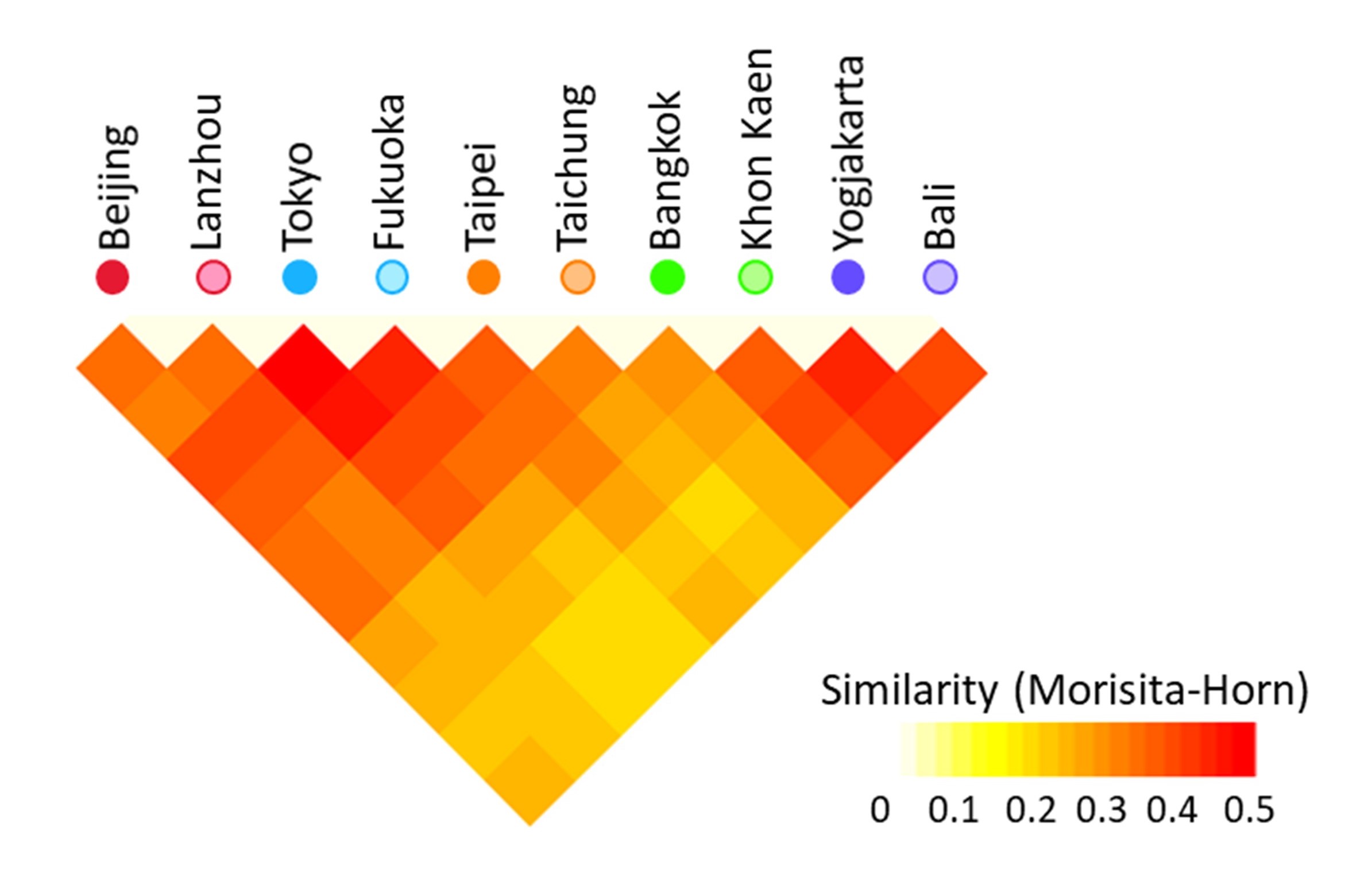
Fig.4. Similarity of gut microbiota among children from 10 cities of 5 countries in Asia.
Reddish color represents high similarity in gut microbiota between children from cities. Gut microbiota of children in Tokyo and Fukuoka showed higher similarity compared to two cities in other countries, suggesting monotonous microbiota in Japanese children.
Furthermore, we performed a predictive metagenomic analysis called "PICRUSt" to address the features of functionality of each type of microbiota.
As a result, it was suggested that P-type microbiota is more abundant in alpha-amylase gene, while BB-type microbiota is more abundant in alpha-glucosidase involved in digestion of oligosaccharides. (Fig.5)
On the other hand, BB-type microbiota is more abundant in choloylglycine hydrolase gene involved in deconjugation of bile acids, compared to P-type microbiota, suggesting higher activity in bile salt metabolism in BB-type microbiota.
It is known that Prevotella abundant in P-type microbiota is sensitive to bile acids.
Further, abundance of choloylglycine hydrolase gene negatively correlates with the number of observed OTUs, suggesting the gut microenvironment not allowing the colonization of a portion of bacteria group in BB-type microbiota.
We are interested in the hidden factor generating P-type and BB-type microbiota, each having distinct structure and function.
Food factors, notably staple food or its cooking style, or whole dietary habit are supposed to determine this kind of global enterotypes.
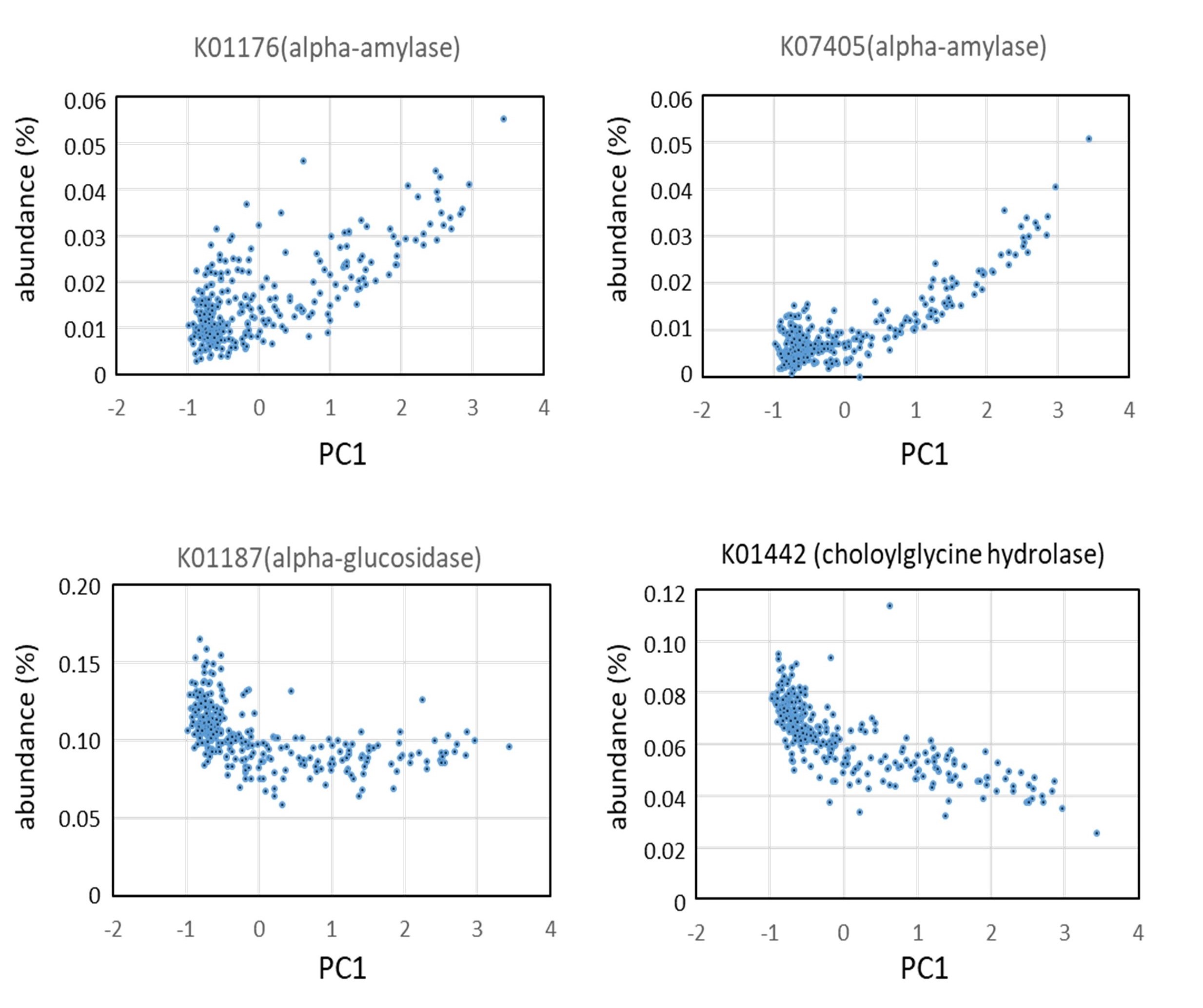
Fig.5. Correlation between abundance of glycosidase gene or genes involved in bile acid metabolism and PC1 values in the principal component analysis of the Asian children.
Abundance of gene of starch-degrading enzyme, amylase, positively correlates with PC1, suggesting high activity of starch degradation in P-type microbiota. On the other hand, choloylglycine hydrolase negatively correaltes with PC1, suggesting high metabolic activity for bile acids in BB-type microbiota. The results suggest that P-type microbiota was established in the digestive tract of children who consume high ratio of crops, particularly Indica rice containg high ratio of indigestible starch, while BB-type microbiota is colonized in the digestive tract of children who consume high-fat diet.
Phase-II study: Construction of basal database of Asian microbiome (Paper in preparation)
Here we aim to build up basal database of microbiome of Asians from newborns to elderlies based on the data collected in AMP. Regarding toddlar, we have already collected the 16S rRNA composition data of fecal samples of 118 Indonesian aged from 0 y to 4 y and 202 Japanese aged from 0 y to 1 y. We also collected whole shot-gun metagenomic data of part of the samples. Regarding analysis from school-age children to elderlies, we have analyzed Korean and Mongolian samples in addition to the samples from five countries in Phase-I study. Thus far, we have analyzed 16S rRNA gene seuqnece of more than 500 hundreds of samples and found common trends in decrease of Bifidobacterium and increase of Enterobacteriaceae with age. Although this trend has been reported in many other studies, it is important to confirm it as commond trend in these Asian countries. However, this trend includes a large variance among individuals and a large number of outliers. By focussing on these samples, we are supposed to address the cause and effect of the decreae of Bifidobacterium and increase of Enterobacteriaceae on the individual health and disease.
Phase-III study (1): Study on foods and gut microbiome of children on Leyte island in Philippines
[Paper published in Frontiers in Microbiology, 2017, 8, 197(ref.2)]
In Phase-III study, we focused on the relationship between foods and gut microbiota and performed a cross-sectional study in certain areas. Here we investigated gut microbiome of Filipino children living Leyte island that is the 8th biggest island under developing in Philippines. We collected fecal samples and dietary records from children living in two cities, namely Ormoc in industrialized site and Baybay in rural site(Fig.6A). The 16S rRNA gene amplicon analysis indicated that Baybay children mostly harbored P-type microbiota, while Ormoc children mostly harbors BB-type microbiota. According to the dietart records, Ormoc children consumed Westernized foods such as much more amount of meats, snacks, and fast foods compared to Baybay children(Fig.6B). Estimated ratio of consumed macronutrients indicated that the energy ratio of consumed fat in Ormoc children was 27.2% compared to 18.1% of Baybay children(Fig.6C). Actually, there were a number of obese children taking fat more than 30% of total calorie, that is the upper limit of fat consumption ratio recommended by WHO. The fat consumption ratio positively correlated with Firmicutes/Bacteroidetes(F/B) ratio and negative correlated with the abundance of Prevotella(Fig.7). The result of PICRUSt analysis indicated enrichment of genes involved in bile acid metabolism in the BB-type microbiota compared to P-type microbiota, sugesting introduction of higher amount of fat into BB-type gut microbiota as suggested by the Phase-I study.
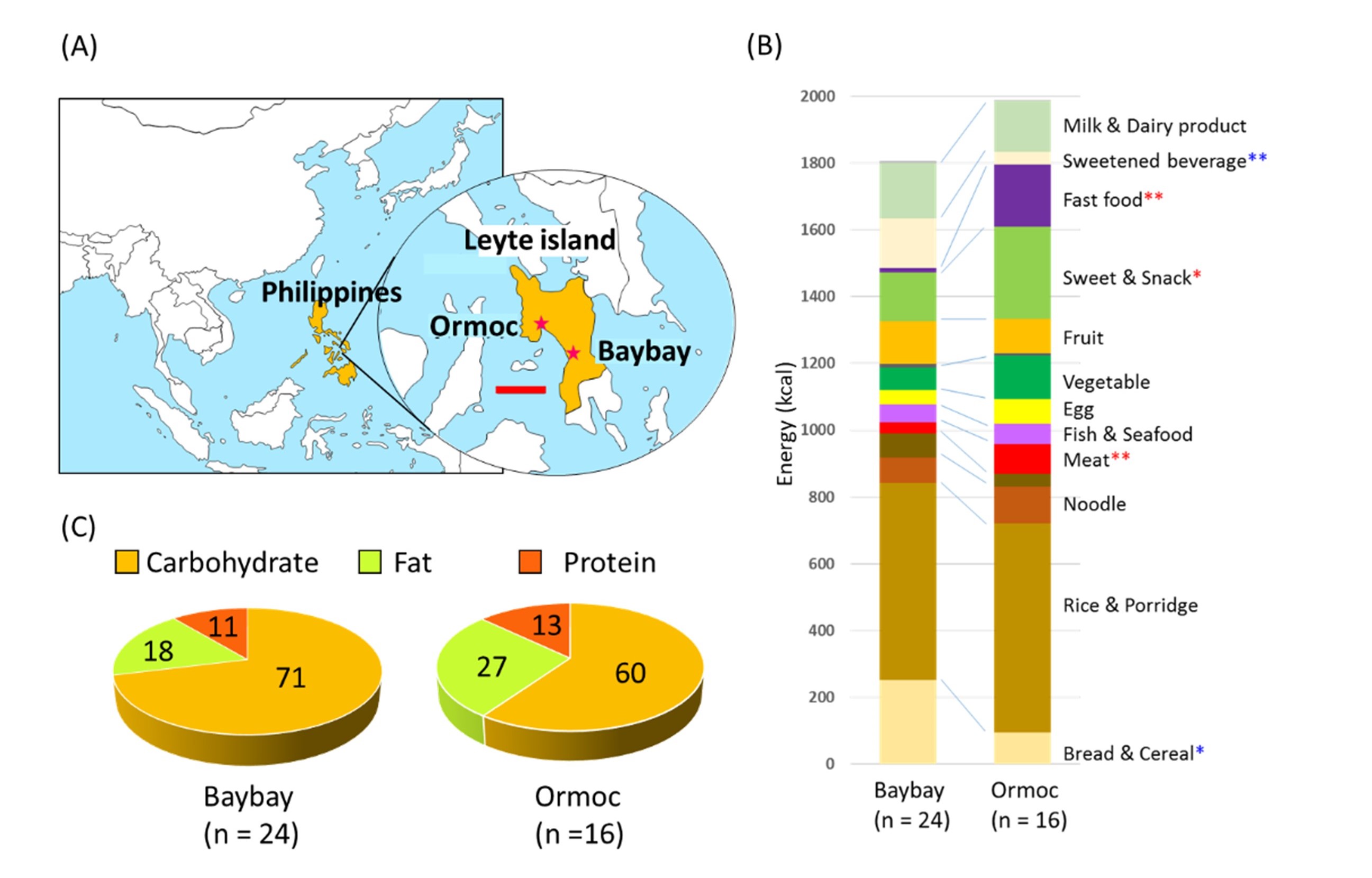
(A)Map indicating position of sampling points, namely Ormoc and Baybay cities.(B)Composition of food energy sources in children in the two cities.(C)Consumed macronutrient ratio of children in the two cities. The data indicates that the children in the urban Ormoc city tend to consume modern Western diet containing more fat compared to the children in the rural Baybay city.
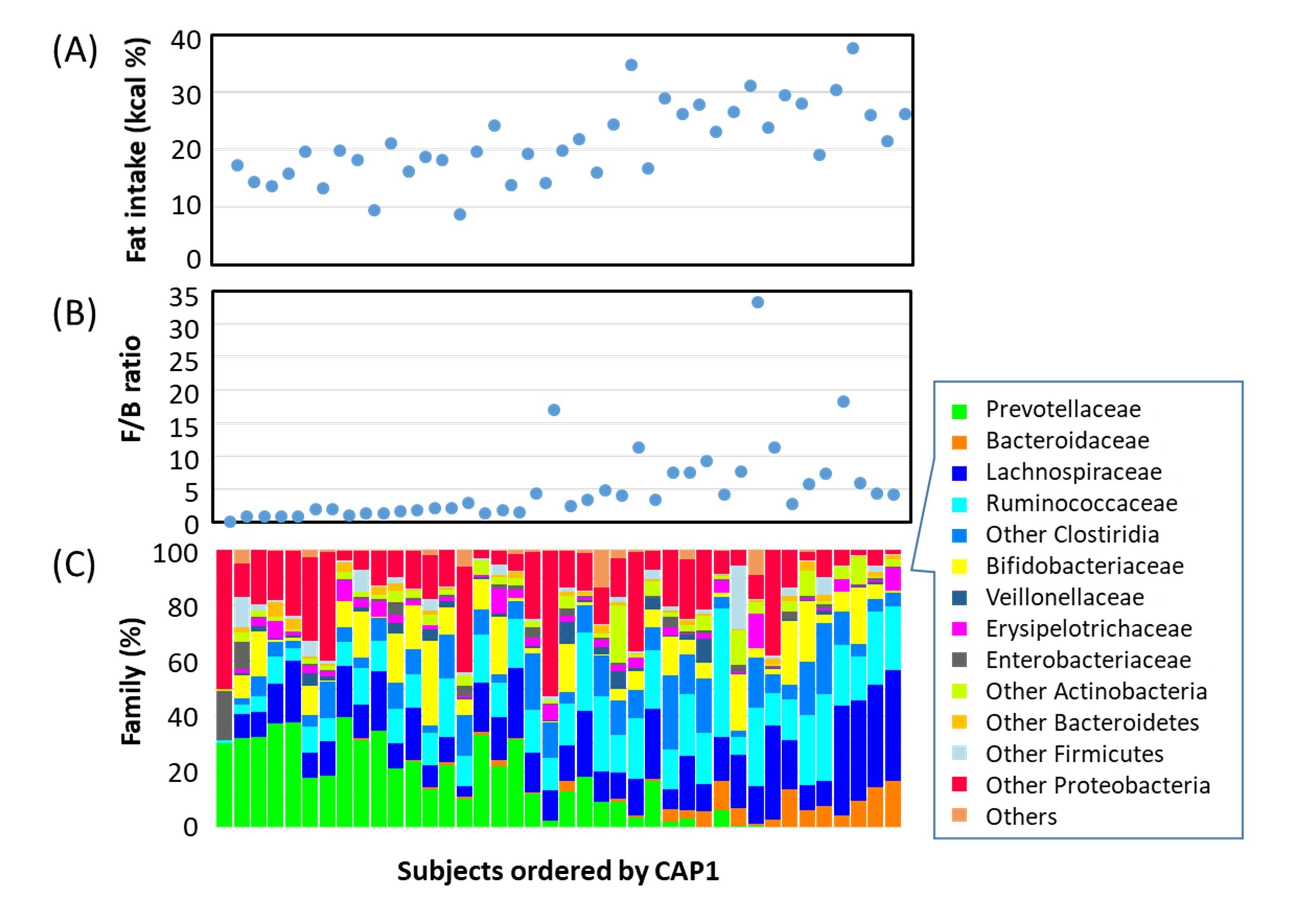
Fig.7.Correlation between fat consumption and gut microbiome in the children on Leyte island.(A)The amount of consumed fat.(B)Firmicutes(F)to Bacteroidetes (B)ratio.(C)Bacteria composition at family level.
Subjects in left side who tend to consume low amount of fat carry low F/B ratio microbiome, notably high amount of Prevotella (Green). On the other hand, subject in right side who consume high amount of fat carry high F/B ratio microbiome, particularly high amount of Lachnospiraceae (blue), Ruminococcaceae (sky blue), and Bacteroidaceae (orange).
Phase-III study (2): Investigation of foods and gut microbiota in school-age children in Thailand.
[Paper published in Frontiers in Microbiology, 2018; 9: 1345 (ref.3)]
Following the above study in Leyte island in Philippines, we investigated gut microbiota of school-age children in Thailand.
The investigation was performed in highly developed city, Bangkok, and Buriram where the people still maintain Thai traditional lifestyle.
Buriram is located in north-east part of Thailand called Isaan region having a representative traditional culture in Thailand.
Particulary, there is a unique food culture represented by "Somtan (Papya salad)", "Sticky rice", "Pla-ra (fermented fish)", and "Nham (fermented pork)" in Isaan region.
In this study, it was confirmed that children in Buriram consume much more amount of vegetable compared to Bangkok children and lower amount of fat (Fig.8).
We also analyzed fecal metabolome as well as microbiome and found that consumed amount of fat negatively correlated with alpha diversity of gut microbiota and also short chain fatty acid concentration in feces (Fig.9).
It is known that short chain fatty acids are main fermentative products of gut microbes which have hormonal functions to control host metabolic and immunological homeostasis and the their lack
causes many disease, notably lifestyle disases such as obesity and type 2 diaetes.
Indeed, Bangkok children short in the production of short chain fatty acids tended to show the BMI in the range of obese or overweight range.
This finding indicates that gut microbiota in Bangkok children living in the city introduced urban Western foods has been remodeled and tends to loose the authentic functionality.
It may warn the crisis of Asian children living in highly developing area.
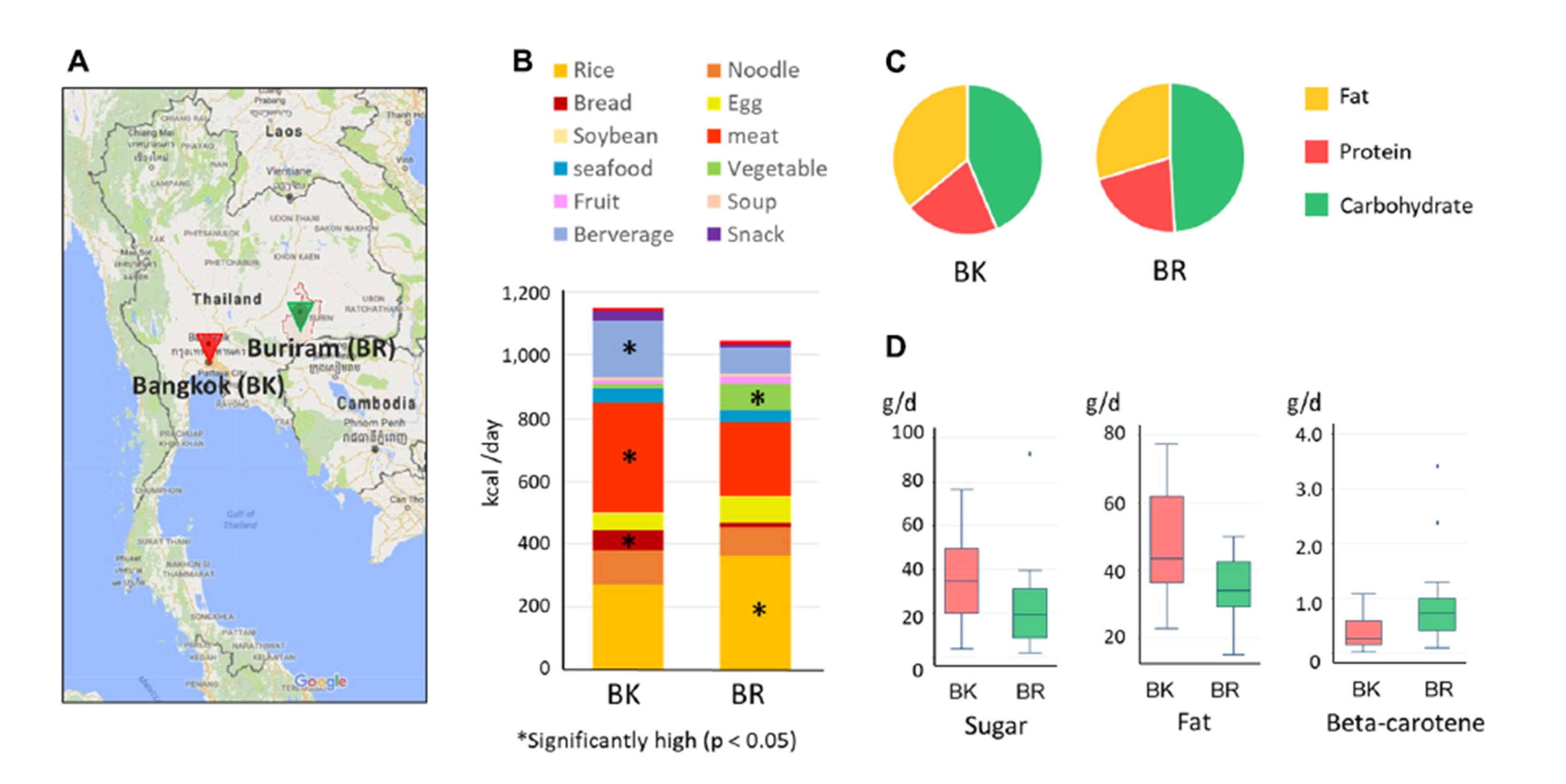
Fig.8. Investigation of dietary habit of children living in uraban city, Bangkok, and rural city, Buriram, in Thailand.
(A)Map indicating the position of sampling cities.(B)Composition of food energy sources in children in the two cities.(C)Consumed macronutrient ratio of children in the two cities.(D)Nutrients showing significant difference in the consumed amount between the two cities. Children in rural city, Buriram, tend to have dietary habit consuming significantly higher amount of vegetables compared to children in Bangkok.
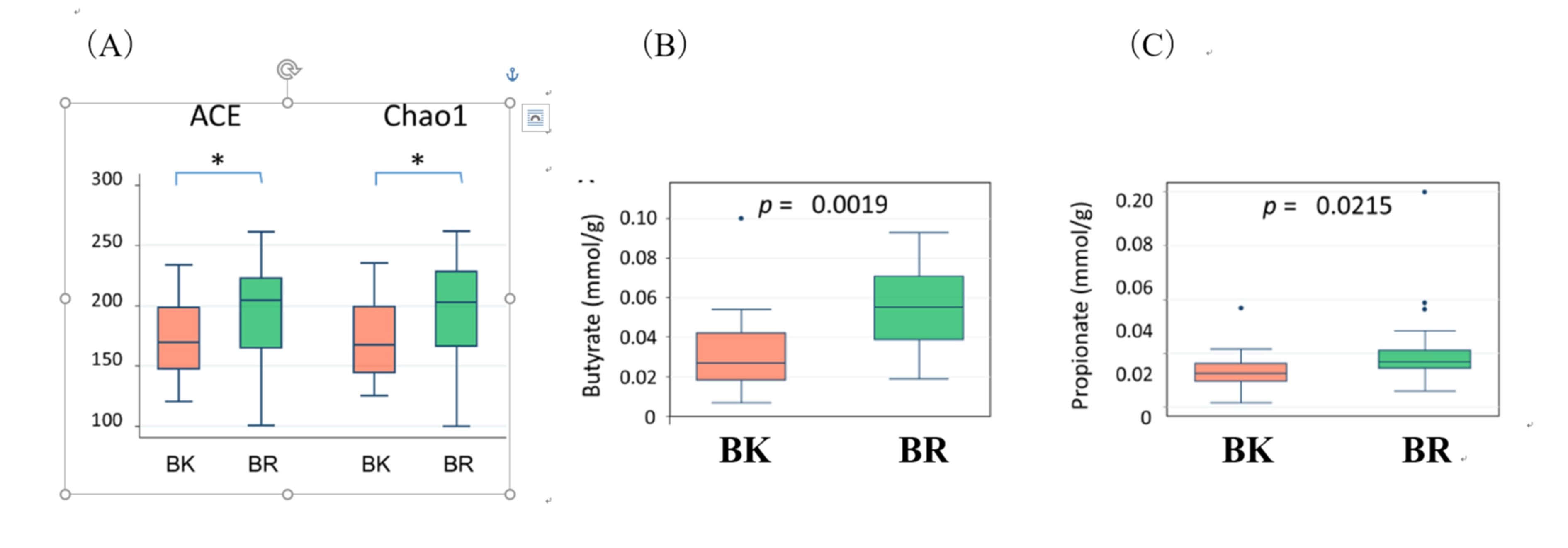
Fig.9. Alpha diverity of gut microbiota (A) and fecal concentration of short chain fatty acids (Butyrate(B) and propionate(C)) in Bangkok children (BK) and Buriram children (BR)。
(A)Gut microbiota of Bangkok children is short in alpha diversity and short chain fatty acid compared to Buriram children. Because short chain fatty acids have indispensable function for host health, we need to keep an eye on the gut microbiota of Bnagkok children.

Fig.8. Investigation of dietary habit of children living in uraban city, Bangkok, and rural city, Buriram, in Thailand.
(A)Map indicating the position of sampling cities.(B)Composition of food energy sources in children in the two cities.(C)Consumed macronutrient ratio of children in the two cities.(D)Nutrients showing significant difference in the consumed amount between the two cities. Children in rural city, Buriram, tend to have dietary habit consuming significantly higher amount of vegetables compared to children in Bangkok.

Fig.9. Alpha diverity of gut microbiota (A) and fecal concentration of short chain fatty acids (Butyrate(B) and propionate(C)) in Bangkok children (BK) and Buriram children (BR)。
(A)Gut microbiota of Bangkok children is short in alpha diversity and short chain fatty acid compared to Buriram children. Because short chain fatty acids have indispensable function for host health, we need to keep an eye on the gut microbiota of Bnagkok children.
Phase-IV study: The link of gut microbiota altered by high-fat modern diet to lifestyle diseases such as obesity and type 2 diabetes
[Paper published in Microorganisms, 2021, 9(5):897(ref.4) and FEMS Microbiol. Letters, 2021, 368(20), fnab149 (ref.5)]
In the Phase-IV study of AMP, we are interested in the fact that modern high fat diet alters gut microbiota, which may predispose people to certain lifestyle diseases, notably metabolic diseases such as obesity and type 2 diabetes. To address this notion, we have performed two cross-sectional studies in Indonesia and Phippines, in which fecal microbiome and metabolome were investigated in relation to obesity and type 2 diabetes. In the study in Indonesia, we found that distinct aspect in obese patients and T2D patients. In obese subjects, gut microbiome showed less alpha diversity with loss of wide-range of commensal groups, suggesting dysbiosis of gut microbiota which was likely due to high fat intake. On the other hand, T2D patients showed higher colonization of specific taxonomic group that is Bacteroides fragilis. The high abundance of B. fragilis associated with depletion of conjugated bile acids, particularly tauroursodeoxy cholic acid (TUDCA) and glycoursocholic acid (GUDCA) which are known to play an important role for the control of blood glucose level (Fig. 10). Furthermore, we found that anti-diabetic drug, metformin, decreased the colonization of B. fragilis and recovered the level of conjugated bile acids. This is a recently discovered new mode of action of metformin (L. Sun et al., 2018, Nature Medicine, 2018, 24, 1919-1929). It is interesting to observe this new aspect of metformin in the cross-sectional study in Indonesia.

Fig.10. Distribution of the key bacteria and anti-diabetic UDCAs among Indonesian subjects. (A) Comparison of abundance of the key taxa among non-T2D, metformin-treated T2D, and non-treated T2D groups. (B) Relative abundance of genera Prevotella and Bacteroides and B. fragilis (upper graph) and GUDCA, TUDCA, and UDCA (lower graph) in our 75 Indonesian subjects ordered by the FBG level. Red line indicates fasting blood glucose. Red asterisks above the graph indicate subjects administered metformin. Blue crosses in the bottom graph indicate the samples lacking in the bile acid data. (Figure cited from ref.4).
In the study in Philippines, we collected fecal samples in uraban city, Manila, and rural site, Albay. In Albay, majority of gut microbiota is Prevotella type, while in Manila, Bacteroides-Firmicute type microbiota were main. There were less T2D patients in Prevotella-type carriers, while Bacteroides-Firmicute microbiota carriers include higher ratio of T2D patients (Fig. 11). The shift from Prevotella type to Bacteroides-Firmicute type is associated with non-traditional less carbohydrate diets. The shift from Prevotella type are commonly observed in developing area all over the world. This kind of global change in gut microbiota should be kept an eye in microbiological science as well as human science.

Fig.11.Urban-type low-carbohydrate diets restructured the Prevotella-driven gut microbiome, resulting to predispose Filipino people with high energy diet to type 2 diabetes mellitus(Figure cited from ref.5).
PUBLICATIONS
Original Papers
[ref.1] Nakayama, J., Watanabe, K., Jiang, J., Matsuda, K., Chao, S.-H., Haryono, P., La-ongkham, O., Sarwoko, M.-A., Sujaya, I, I. N., Zhao, L., Chen, K.-T., Chen, Y.-P., Chiu, H.-H., Hidaka, T., Huang, N.-X., Kiyohara, C., Kurakawa, T., Sakamoto, N., Sonomoto, K., Tashiro, K., Tsuji, H., Chen, M.-J., Leelavatcharamas, V., Liao, C.-C., Nitisinprasert, S., Rahayu, E. S., Ren, F.-Z., Tsai, Y.-C., Lee, Y.-K*. Diversity in gut bacterial community of school-age children in Asia. Sci Rep., 2015, 5, 8397.[ref.2] Nakayama, J.*, Yamamoto, A., Palermo-Conde, L. A., Higashi, K., Sonomoto, K., Tan, J., Lee, Y.-K. Impact of westernized diet on gut microbiota in children on Leyte island., Front Microbiol., 2017, 8, 197.
[ref.3] Kisuse, J. La-ongkham, O., Nakphaichit, M., Therdtatha, P., Momoda, R., Tanaka,M., Fukuda,S. Popluechai, S., Kespechara, K., Sonomoto, K., Lee, Y.-K., Nitisinprasert, S., Nakayama, J.* Urban diets linked to gut microbiome and metabolome alterations in children: a comparative cross-sectional study in Thailand. Front. Microbiol., 2018, 9, 1345.
[ref.4] Therdtatha P, Song Y, Tanaka M, Mariyatun M, Almunifah M, Manurung NEP, Indriarsih S, Lu Y, Nagata K, Fukami K, Ikeda T, Lee YK, Rahayu ES, Nakayama J. Gut Microbiome of Indonesian Adults Associated with Obesity and Type 2 Diabetes: A Cross-Sectional Study in an Asian City, Yogyakarta. Microorganisms. 2021, 9(5):897. doi: 10.3390/microorganisms9050897.
[ref.5] Watanabe, M., Sianoya A., Mishima, R., Therdtatha, P., Rodriguez, A., Ramos, D. C., Lee Y.-K., Dalmacio, L. M., Nakayama, J. Gut microbiome status of urban and rural Filipino adults in relation to diet and metabolic disorders., FEMS Microbiol.Letters, 2021, 368(20), fnab149, https://doi.org/10.1093/femsle/fnab149.